AMF Medical SA bought for CHF 200 million by Tandem Diabetes Care
Share this article
Tandem Diabetes Care, a global US-Based insulin delivery and diabetes technology company, announced an agreement to acquire AMF Medical SA, the privately held Swiss developer of the Sigi™ Patch Pump based at EPFL Innovation Park in Lausanne. The full agreement could reach CHF 200 million but is subject to future technical developments and FDA approval
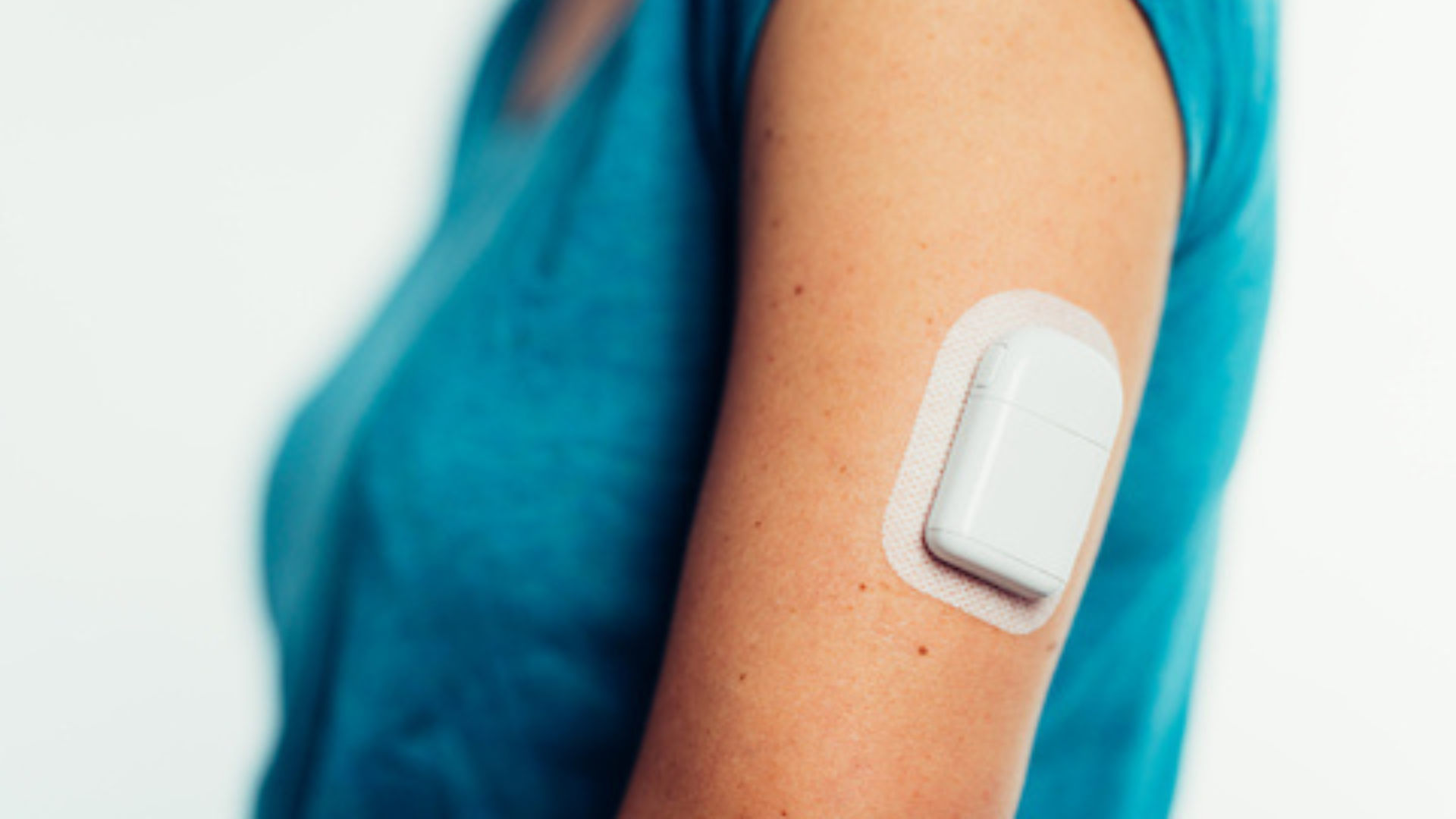
The Sigi Patch Pump, created by AMF Medical SA, is under development and not commercially available. It is designed to be an ergonomic, rechargeable patch pump that reduces the burden of managing diabetes through its use of pre-filled insulin cartridges and its compatibility with automated insulin delivery technology.
“This acquisition supports our commitment to drive growth through innovation, as AMF Medical has novel technology and a shared passion for creating easy-to-use solutions to improve the lives of people with diabetes,” said John Sheridan, Tandem Diabetes Care president and chief executive officer. “Our portfolio approach to diabetes care is designed to bring the benefits of advanced insulin pump therapy to more people worldwide, utilize our expertise in automated insulin delivery technology and data management, and leverage our commercial infrastructure to create shareholder value.”
AMF Medical’s proprietary and disruptive insulin delivery solution is expected to:
- Expand Tandem’s type 1 and type 2 addressable market opportunities by adding a pump designed for a segment of people living with diabetes not served by Tandem today;
- Accelerate time-to-market for a patch pump in Tandem’s portfolio of technology offerings;
- Further Tandem’s sustainability objectives by offering an additional pump that minimizes the generation of electronic and battery waste;
- Provide a pathway for channel expansion and increased customer access.
Tandem Diabetes Care will acquire AMF Medical under the following financial terms of the agreement:
- A previous strategic investment of Swiss Francs (“CHF”) 8.0 million paid in the third quarter of 2022
- A cash payment of CHF 62.4 million due at closing
- Additional contingent earnout payments of up to CHF 129.6 million, in aggregate, payable upon the achievement of certain milestones, including:
- Up to CHF 38.4 million upon the successful completion of key development milestones over the next two years
- Up to CHF 91.2 million upon obtaining regulatory clearance of an automated controller enabled (ACE) pump by the United States Food and Drug Administration
The transaction is subject to the satisfaction of customary closing conditions and is expected to close in January 2023.
Source: Press Release