Business Opportunities
If you would like your business opportunities to be listed here, please fill in the form to upload your information. Please note, we will only post opportunities related to life sciences.
Latest opportunities
Opportunities
Ilmac – Start-Up Area
Start-ups and newcomers are welcome to participate at Ilmac 2026 in Lausanne (23 – 24 September 2026 – Beaulieu) with…
Opportunities
DayOne Accelerator
Apply before 31 May 2026 for DayOne Accelerator, a programme dedicated to healthtech projects. The DayOne Accelerator is looking for…
Opportunities
SMART Challenge
The SMART Challenge 2026 kicks off on 21 April 2026 —and it needs concrete, innovative ideas to make physical activity…
Opportunities
Venture Technology Connect
Apply for Venture Technology Connect and go to Verbier next November to pitch you project to a selected audience of…
Opportunities
Human Health by Design – Call For Project
Apply before 31 May 2026 for the H4 Human Health by Design programme, and get a 6-month support and up…
Opportunities
Bridge – Proof of Concept
A 12-month programme to develop your idea into a prototype. Apply before 1 June 2026. BRIDGE is a funding programme…
Opportunities
Age Innovation Prize
Apply before 14 June 2026 for the Age Innovation Prize of the Institute for Ageing Research IAF in partnership with…
Opportunities
Healthtech Investor Summit
Apply before 15 June 2026 to pitch at Health Investor Summit (HIS) and show your innovation to +100 life sciences…
Opportunities
Bayer – Co.Lab
Apply before 15 June 2026 for the Bayer Co.Lab programme. Get up to CHF 50,000 and personalised coaching If you’re…
Opportunities
Switzerland – Australia Call For Proposals
Apply before 17 June 2026, the SNSF and the Australia National Health and Medical Research Council (NHMRC) are launching a…
Opportunities
European Academy of Neurology – Start-up Corner
Startups in neurological field can showcase their advancements at the 12th Congress of the European Academy of Neurology (EAN) in…
Opportunities
CAS Personalized Molecular Oncology
Registration is open for the Certificate of Advanced Studies (CAS) in Personalized Molecular Oncology (PMO) — 2026–2027 session! Are you…
Opportunities
Switzerland-South Korea Call For Projects
Swiss and South Korean participants are invited to submit proposals, before 30 June 2026, for joint projects in the research…
Opportunities
IFF Awards
Apply until 30 June 2026 for the IFF (Innovation Fribourg Freibourg) Awards, a initiative of Canton of Fribourg to honor…
Opportunities
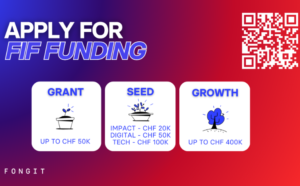
FONGIT Innovation Fund
Send your application before 22 July 2026 for the FONGIT Innovation Fund (FIF) FONGIT, Fondation Genevoise pour l’Innovation Technologique, is…
Opportunities
Future of Health Grant
The Future of Health Grant programme aims to accelerate start-ups with a digital health solution. Apply before 31 July 2026….
Opportunities
Micronarc Prize x PX Group
Apply before 31 August 2026 for the Micronarc Prize x PX Group and try to get CHF 5,000 and a…
Opportunities
Innobooster Gebert Rüf Stiftung
Apply before 1 May 2026 for the Gebert Rüf Stiftung Innobooster and get up to a CHF 150,000 support Gebert…
Opportunities
First Ventures
Apply before 1 September 2026 to First Ventures and get up to CHF 150’000 from Gebert Rüf Stiftung With «First…
Opportunities
FENRIV Research Award
Apply before 1 September 2026 for the Egon Naef Foundation for In Vitro Research’s Research Award The Egon Naef Foundation…
Opportunities
DayOne Accelerator 2.0
Three programmes that enable the growth of global healthcare and medtech startups at various stages of their development Digital Health…
Opportunities
La Forge Incubator at EPFL Innovation Park
Located at the heart of EPFL Innovation Park, La Forge is an incubator for early stage start-ups in the region.
Opportunities
SyNNergy
The Canton of Vaud supports companies working on the digital transition The SyNNergy programme is an initiative of the Canton…
Opportunities
Biopôle Vanguard Accelerator
This six-month programme, supported by Biopôle SA and its partners, helps health-oriented projects to grow. The Vanguard Accelerator is a…
Opportunities
Debiopharm invests in Digital Health
Debiopharm’s strategic corporate fund invests in digital health, smart data & innovative tech start-ups The fund typically invests in…
Opportunities
Biopôle Startup Fund
Up to CHF 90,000 of support per year for up to three consecutive years to help life sciences companies with…
Opportunities
Innosuisse: Innovation for startups
Competitive project funding for start-ups before market entry Innosuisse supports science-based projects of start-ups with significant innovation potential. These innovation…
Opportunities
CERN Venture Connect
The CERN Venture Connect (CVC) programme aims to act as a connector to bring together great entrepreneurs, provide them with…
Opportunities
GENILEM
Since 1995, GENILEM helps entrepreneurs in Vaud and Geneva to build innovative and responsible businesses GENILEM supports innovative entrepreneurs,…
Opportunities
FIT – Fondation pour l’Innovation Technologique
The Foundation for Technological Innovation offers grants and loans to support the development of innovative technological projects at various stages…
Opportunities
Lichtsteiner Foundation
Try to be one of the 10 early-stage healthcare startups supported by the Lichtsteiner Foundation each year. Application accepted throughout…