Artiria Medical successfully secures USD 6 Million
Share this article
Artiria Medical secures funding to complete its clinical studies in stroke in both Europe and the Us, following promising first in-human results and FDA clearance.
The MedTech company, developing next-generation neuro-interventional products for stroke and aneurysms, successfully closed off a Series A2 round of USD 0.6 Million. Furthermore, they secured an additional USD 600,000 FIT Tech Growth loan.
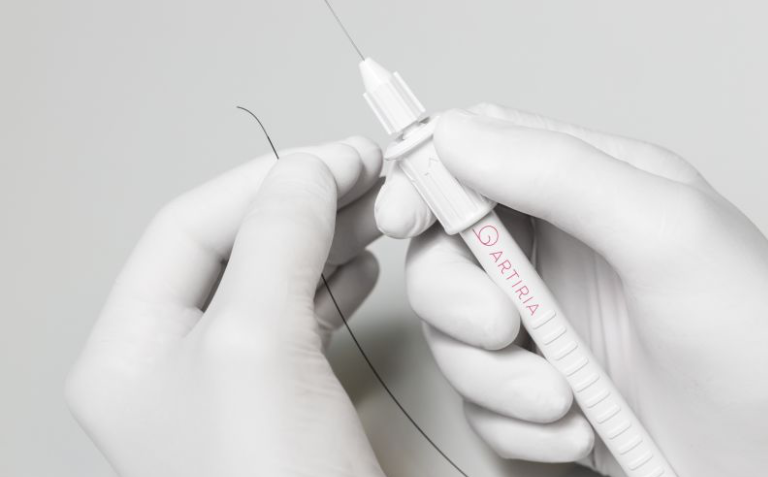
Artiria’s leading product is a novel real-time deflectable guidewire that enables Neurointerventional surgeons to access the diseased site faster. The product has already been used in humans and treated its first patients in 2023.
This Series A2 investment round, led by 4FOX Ventures, was joined by the Wyss Center for Bio and Neuroengineering. Existing investors include Verve Ventures, the Zürcher Kantonalbank, NEST pension fund, and business angels.
“This funding round shall enable Artiria to study the unique features of our product with key opinion leaders in both Europe and the US through a randomised controlled study against the market-leading products.” said Guillaume Petit-Pierre, CEO and co-founder of Artiria. “Following the already secured FDA clearance, this study will further enable Artiria to apply for European market approval thus significantly extending the scope of the company.”
Valentin Chenaux General Partner of 4FOX Ventures commented, “We believe that Artiria’s innovative solution will advance the treatment of stroke with a product enabling safe yet accelerated procedures and we are delighted to be part of this great journey.”
Source: press release